A new method to find modulators of the immune response to COVID-19
The recent paper, published in Science Advances by the Computational Biology Laboratory at CICbioGUNE, member of the Basque Research & Technology Alliance BRTA and at Luxembourg Centre for Systems Biomedicine (LCSB) in Luxemburg, describes a new method of how molecules can be identified that amplify and maintain the inflammatory response upon an infection
They use their novel method to identify these molecules in COVID-19 patients and suggest new potential targets for therapeutic intervention in severe cases of COVID-19
 Upon getting infected for instance by a virus, the human immune system is typically triggered to fight the intruder. The immune response is composed of complex processes and involves various types of cells, which communicate with each other via so-called cytokines.
Upon getting infected for instance by a virus, the human immune system is typically triggered to fight the intruder. The immune response is composed of complex processes and involves various types of cells, which communicate with each other via so-called cytokines.
A major task of the immune system is to identify and remove those cells of the body that got infected which causes inflammation.
“Normally, the immune system is tightly controlled keeping the tissue damage as low as possible,” explains Antonio del Sol group leadr at Computational Biology Laboratory in CIC bioGUNE, “However, in conditions where the pathogenic factor cannot be readily cleared, we know from other infectious diseases that patients could develop a hyperinflammatory condition where the immune response is amplified and maintained by positive feedback loops. This hyperinflammatory condition increases the severity of the symptoms and might even lead to death. The present challenge is how to alleviate inflammation while not lowering the patient’s ability to clear SARS-CoV-2, the virus causing COVID-19”, he continues.
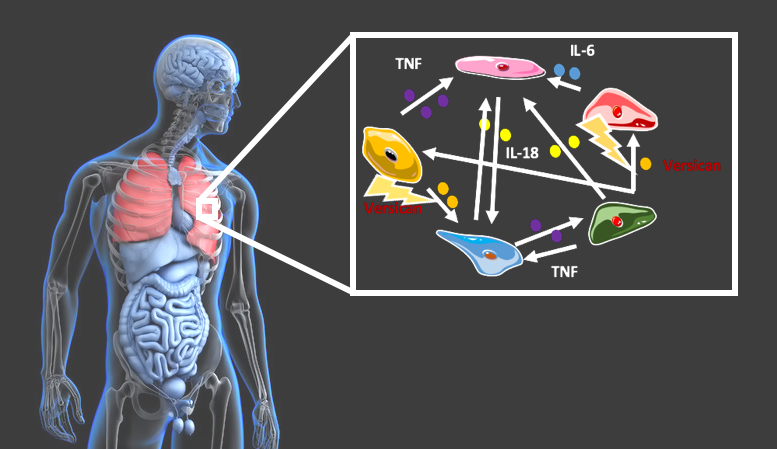
To address this problem, the researchers developed a method by which modulators of the immune response can be identified. Knowing the molecules amplifying the inflammation, their activity could be brought back to innocuous levels.
Analysing more than 1,700 cell-cell interactions, the group created a comprehensive map of the immune response in the lungs of COVID-19 patients with mild and severe symptoms. The special characteristic of this approach is that the genomic information of individual cells was used to create these maps, thus representing also the different cell types of the immune system.
“We decided to use our model in the context of COVID-19 as patients with severe disease courses also often exhibit hyperinflammation putting their life in danger,” Sascha Jung first author of the publication. “We found that the increased inflammation is caused by feedback mechanisms, which also partially impair the immune cells and thus exacerbate the inflammatory response,” he adds.
Del Sol and his team used their model to identify those molecules that might be able to modulate the runaway inflammatory response of the immune system. He summarizes the findings of this approach: “By computational perturbation of genes involved in the feedback loops, we predicted the proteins Versican and the Toll-like Receptor 2 to play a major role. According to our model, an inhibition of either of these molecules can disrupt up to 75% of the feedback loops without interfering with the general immune response.”
The protein Versican resides in the extracellular matrix and is involved in several intercellular communication processes. In turn, the Toll-like Receptor 2 resides at the surface of cells where it detects pathogenic molecules. Both proteins are known to play a role in the human immune response.
The results of this study have already attracted the attention of immunologists at the “Cytokines” conference, the world’s most important conference on basic, translational and clinical research related to cytokine biology, where Del Sol’s work has been presented. According to these novel findings, the predicted proteins constitute a potential target for medical intervention to treat COVID-19, which is currently being evaluated in animal models together with immunologists from the University of Washington, USA.
The full publication can be found here:
https://advances.sciencemag.org/content/7/6/eabe5735/tab-article-info




